What Is Chronic Inflammation — and Why an Anti-Inflammatory Diet Is Part of Modern Medicine
Inflammation is one of the body’s most powerful defense mechanisms. But when it fails to switch off, it becomes one of the most destructive forces in the human body — quietly driving heart disease, cancer, and cognitive decline for years before any symptoms appear. Understanding how chronic inflammation works is the first step toward taking it seriously. And increasingly, an anti-inflammatory diet is recognized not as a wellness trend, but as a legitimate clinical tool.
What Is Inflammation — and When Does It Become a Problem?
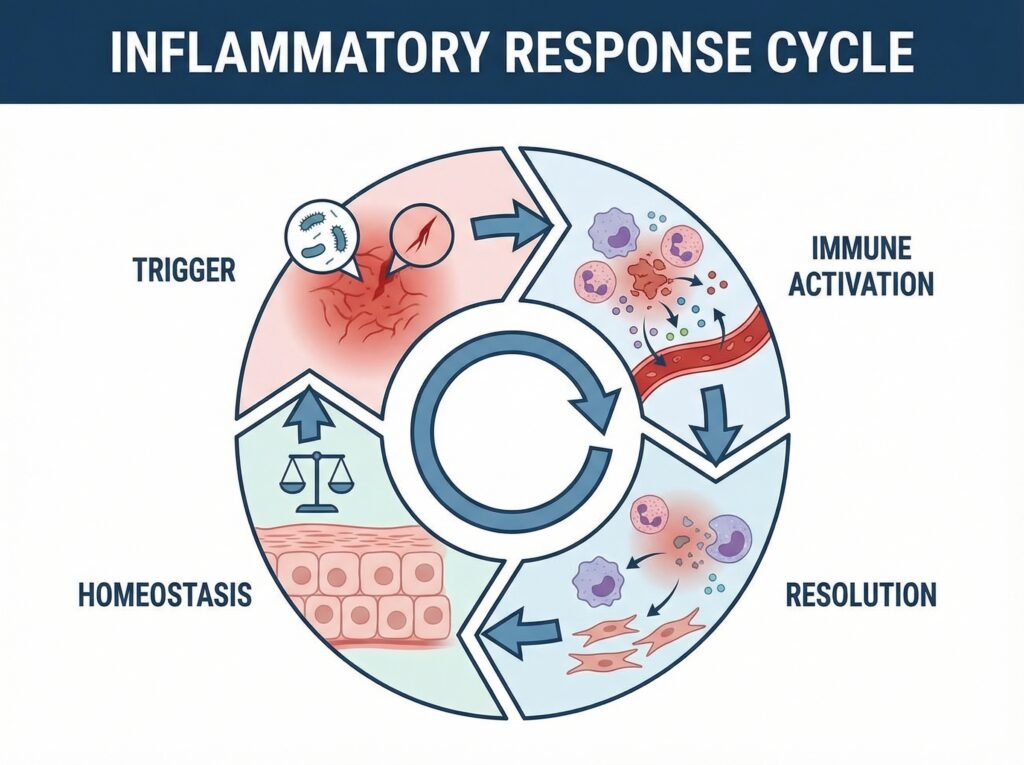
The inflammatory response is crucial to human survival. Inflammation is a normal and vital reaction to internal injury and external threats — whether that means a bacterial infection, a physical wound, or a toxic substance entering the body.
When regulated properly, the inflammatory response does three things: it eliminates the harmful agent, repairs damaged tissue, and returns the body to a state of balance known as homeostasis.
The problem begins when this process fails to stop.
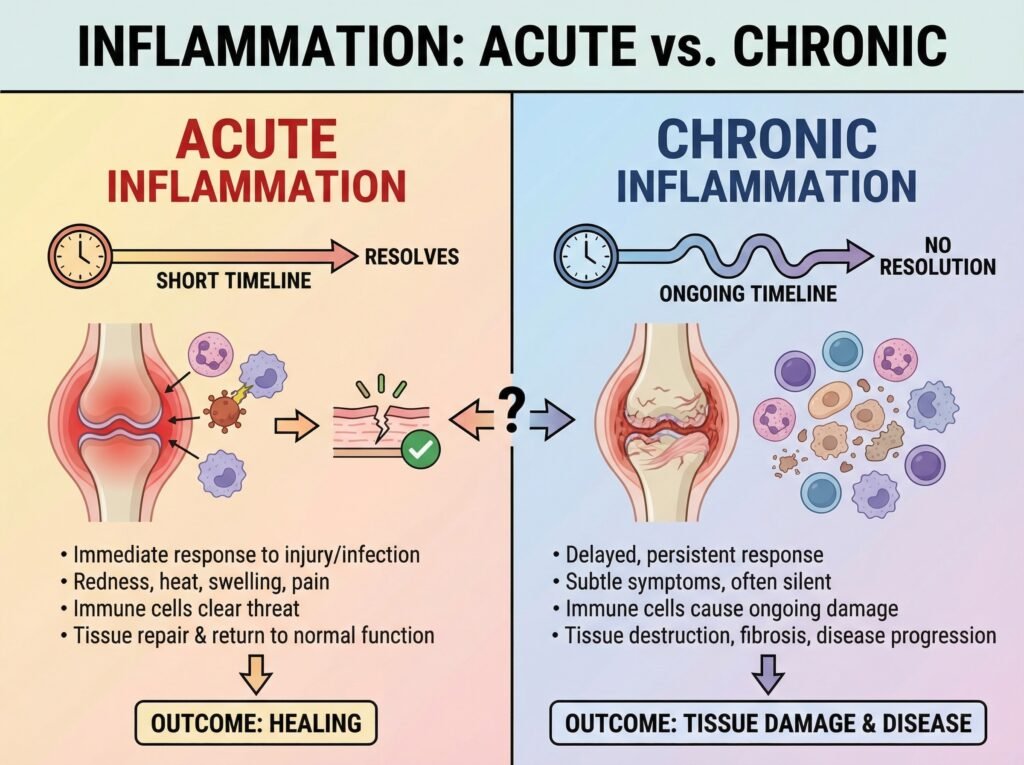
Acute vs. Chronic Inflammation: A Critical Difference
Acute inflammation is the familiar kind — fast, intense, and short-lived. It begins within minutes to hours, lasts for hours to days, and is initiated by tissue-resident macrophages and dendritic cells. In response to a perceived threat, these cells release a cascade of signaling molecules: proinflammatory cytokines, chemokines, and prostaglandin E2 (PGE2).
This process has three defining characteristics:
- Enhanced blood flow to the affected area through dilation of small blood vessels
- Increased vascular permeability, allowing immune cells to reach the site quickly
- Migration of phagocytic leukocytes — white blood cells that engulf and destroy pathogens — into the affected tissue
Think of it as an emergency response team: it arrives fast, does its job, and leaves. A sprained ankle swells and becomes tender for a few days, then heals. That is acute inflammation working correctly.
Chronic inflammation operates on an entirely different timescale — and with entirely different consequences. It develops when the acute response cannot resolve normally, either because the triggering stimulus persists or because the body’s regulatory mechanisms break down.
The result: proinflammatory processes continue unchecked by anti-inflammatory processes. The body stays in a state of low-level internal assault, damaging its own tissues day after day.
The Mechanism: Why Chronic Inflammation Damages Healthy Tissue
A key and often overlooked fact: even in a normal, well-functioning acute inflammatory response, leukocytes are important causes of injury to normal cells and tissues. Their destructive capacity — essential for killing pathogens — does not discriminate perfectly between foreign invaders and host tissue.
In acute inflammation, this collateral damage is temporary and acceptable. The immune system resolves the response before lasting harm occurs.
In chronic inflammation, there is no resolution. The triggering factors — free radicals, oxidative stress, persistent pathogens — continue to activate the inflammatory machinery. The damage accumulates. Over time, this produces local or systemic organ damage that underlies some of the most serious noncommunicable diseases of our time.
Three Major Diseases Driven by Chronic Inflammation
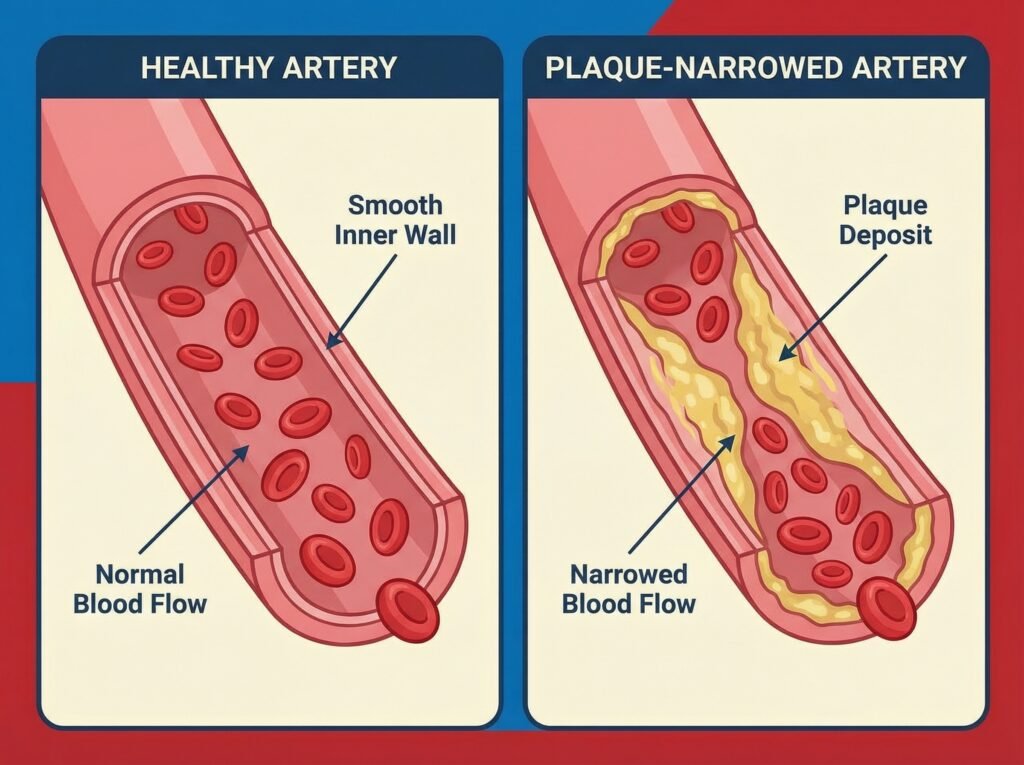
1. Cardiovascular Disease and Atherosclerosis
Atherosclerosis — the buildup of plaques within arterial walls — is not simply a mechanical problem of fat accumulating in blood vessels. It is a chronic inflammatory response within the arterial wall to ongoing endothelial injury.
Macrophages accumulate inside the vessel wall, become chronically activated, and release proinflammatory cytokines. They recruit additional immune cells to the site, exert a catabolic effect on fibrous atheromatous plaques, and progressively increase the risk of plaque rupture and thrombosis — the events that trigger heart attacks and strokes.
Cardiovascular disease remains the most common underlying cause of death in the United States. Chronic inflammation is central to its pathogenesis.
2. Cancer
Cancer’s relationship with chronic inflammation is bidirectional and particularly complex. Cancer cells express antigens that the immune system can recognize, which upregulates proinflammatory cytokines and triggers ongoing immune activation. Cancer cells also frequently undergo necrosis, promoting a continuous influx of leukocytes into the tumor environment.
At the same time, cancer cells possess the ability to evade normal immune destruction while selectively promoting immune responses that support their own growth. The resulting inflammation is dysregulated and dysfunctional — it does not eliminate the tumor but actively facilitates its progression [5].
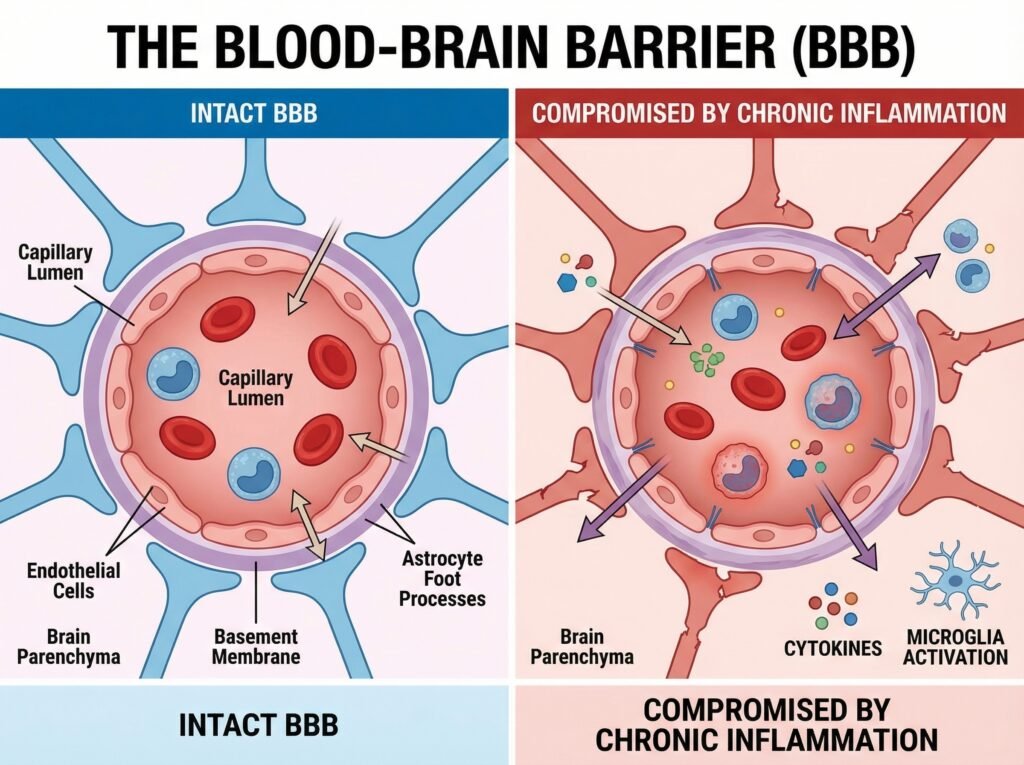
3. Neurocognitive Decline
The brain was long thought to be largely insulated from peripheral inflammation by the blood-brain barrier — a selective interface between the central nervous system and the rest of the body. More recent understanding has revised this significantly.
The blood-brain barrier functions as a bidirectional communication system between the brain’s innate immune system and the peripheral immune system. Sustained peripheral inflammation chronically activates microglia — the specialized macrophages of the brain — and promotes a breakdown of the blood-brain barrier itself.
Once compromised, this barrier allows peripheral inflammatory mediators to enter the central nervous system directly, increasing neuroinflammation and elevating the risk of neurocognitive diseases.
Why an Anti-Inflammatory Diet Is Now Part of Medical Practice
Chronic inflammation contributes to the risk of disease development and progression across multiple organ systems. While the full picture is not yet completely understood, the evidence has been substantial enough to shift clinical practice.
Clinicians — particularly primary care practitioners — now incorporate the reduction of inflammation into both preventative and treatment planning. Among the modalities available, adherence to an anti-inflammatory diet is one of the most accessible and consistently recommended interventions.
An anti-inflammatory diet is not a supplement or a prescription. It is a pattern of eating designed to reduce the chronic inflammatory burden on the body — addressing one of the most pervasive drivers of modern chronic disease at its source.
Frequently Asked Questions
Q: What is chronic inflammation? Chronic inflammation is a state in which the body’s inflammatory response continues without resolution, causing ongoing damage to tissues and organs. It occurs when triggering factors such as free radicals, oxidative stress, or persistent pathogens repeatedly activate the immune system beyond its ability to self-regulate.
Q: What diseases are caused by chronic inflammation? Chronic inflammation is a significant contributing factor to cardiovascular disease (atherosclerosis), cancer, and neurocognitive decline, among other noncommunicable conditions.
Q: What is an anti-inflammatory diet? An anti-inflammatory diet is a nutritional approach aimed at reducing the chronic inflammatory burden in the body. It is increasingly recognized by clinicians as a legitimate tool in both preventative medicine and disease management.
Q: How does inflammation damage the brain? Chronic peripheral inflammation can activate microglia — the brain’s immune cells — and break down the blood-brain barrier. This allows inflammatory mediators from the bloodstream to enter the central nervous system, increasing neuroinflammation and the risk of cognitive disease.
Q: Is inflammation always harmful? No. Acute inflammation is a vital and protective response. It becomes harmful when it fails to resolve and transitions into chronic inflammation, which damages healthy tissue over an extended period.
Sources
[1] Pahwa R, Goyal A, Jialal I. Chronic Inflammation. StatPearls Publishing, 2023. https://www.ncbi.nlm.nih.gov/books/NBK493173/
[2] Libby P, Ridker PM, Maseri A. Inflammation and Atherosclerosis. Circulation. 2002;105(9):1135–1143. https://www.ahajournals.org/doi/10.1161/hc0902.104353
[3] Yang S, et al. Research Progress on the Relationship between Atherosclerosis and Inflammation. Biomolecules. 2018;8(3):80. PMID: 30142970 https://pubmed.ncbi.nlm.nih.gov/30142970/
[4] Cai Z, et al. Inflammation in atherosclerosis: pathophysiology and mechanisms. Cell Death & Disease. 2024;15:760. PMID: 39528464 https://pubmed.ncbi.nlm.nih.gov/39528464/
[5] Zhao H, et al. Inflammation and tumor progression: signaling pathways and targeted intervention. Signal Transduct Target Ther. 2021;6(1):263. PMID: 34248142 https://pubmed.ncbi.nlm.nih.gov/34248142/
[6] Coussens LM, Werb Z. Inflammation and Cancer. Nature. 2002;420:860–867. https://pmc.ncbi.nlm.nih.gov/articles/PMC6704802/
[7] Greten FR, Grivennikov SI. Inflammation and Cancer: Triggers, Mechanisms, and Consequences. Immunity. 2019;51(1):27-41. https://www.sciencedirect.com/science/article/abs/pii/S1040842820303139
[8] Vargas-Caballero M, et al. An overview of the relationship between inflammation and cognitive function. Ageing Research Reviews. 2024. https://www.sciencedirect.com/science/article/pii/S0197018624002274
[9] Nation DA, et al. Blood-brain barrier breakdown, neuroinflammation, and cognitive decline in older adults. Alzheimers Dement. 2019;15(2):319. PMID: 30120040 https://pubmed.ncbi.nlm.nih.gov/30120040/
[10] Hartle M, et al. Microglia at the blood brain barrier in health and disease. Front Cell Neurosci. 2024. https://www.frontiersin.org/journals/cellular-neuroscience/articles/10.3389/fncel.2024.1360195/full



